Carlos Martínez-A.
Group Leader
Karel Van Wely
Group Leader
Research summary
With a long scientific career in immunology studying lymphocyte biology and its implications in cell migration and autoimmunity, We now move to the emerging field of stem cell biology, focusing on their role in cancer and immunoregulation. In the last 25 years, tremendous advances have been made in the generation of stem cells from various organisms; this has generated a wealth of knowledge in fields ranging from embryology to development.
Publications
Berzoti-Coelho MG, Ferreira AF, de Souza Nunes N, Pinto MT, Júnior MC, Simões BP, Martínez-A C, Souto EX, Panepucci RA, Covas DT, Kashima S, Castro FA. The expression of Death Inducer-Obliterator (DIDO) variants in Myeloproliferative Neoplasms. Blood Cells Mol Dis 2016; 59: 25-30
Gatchalian J, Gallardo CM, Shinsky SA, Ospina RR, Liendo AM, Krajewski K, Klein BJ, Andrews FH, Strahl BD, M van Wely KH, Kutateladze TG. Chromatin condensation and recruitment of PHD finger proteins to histone H3K4me3 are mutually exclusive. Nucleic Acids Res 2016; 44:6102-12
Rackov G, Hernández-Jiménez E, Shokri R, Carmona-Rodríguez L, Mañes S, Álvarez-Mon M, López-Collazo E, Martínez-A C, Balomenos D. p21 mediates macrophage reprogramming through regulation of p50-p50 NF-κB and IFN-β. p21 mediates macrophage reprogramming through regulation of p50-p50 NF-κB and IFN-β. J Clin Invest 2016; 126:3089–103
Daszkiewicz L, Vázquez-Mateo C, Rackov G, Ballesteros-Tato A, Weber K, Madrigal-Avilés A, Di Pilato M, Fotedar A, Fotedar R, Flores JM, Esteban M, Martínez-A C, Balomenos D. Distinct p21 requirements for regulating normal and self-reactive T cells through IFN-γ production. Sci Rep 2015; 5:7691
Villares R, Gutiérrez J, Fütterer A, Trachana V, Gutiérrez del Burgo F, Martínez-A C. Dido mutations trigger perinatal death, brain abnormalities and behavioural alterations. Proc Natl Acad Sci USA 2015; 112:4803-8
 We study the connections between embryonic stem cell (ESC) differentiation and chromosome instability. Most sporadic tumours have a combination of genetic defects termed chromosomal instability (CIN), found in ~85% of non-hereditary carcinomas. We study aneuploidy (whole-chromosome CIN) and a combination of translocations, duplications and deletions (structural CIN).
We study the connections between embryonic stem cell (ESC) differentiation and chromosome instability. Most sporadic tumours have a combination of genetic defects termed chromosomal instability (CIN), found in ~85% of non-hereditary carcinomas. We study aneuploidy (whole-chromosome CIN) and a combination of translocations, duplications and deletions (structural CIN).
We focus on the Dido locus, a gene complex strongly expressed in ESC, that encodes three splice variants (Dido1, Dido2 and Dido3). Whereas Dido2 and Dido3 have the PHD, TF2SM and SPOC protein domains, Dido1 only encompasses PHD. Dido3, the largest, most broadly expressed isoform, is a nuclear protein that interacts with centrosomes and the synaptonemal complex in somatic and germ cells, respectively.
N-terminal truncation of Dido3 (Dido3ΔNT) provokes centrosome amplification and CIN. Dido3 interacts with several proteins, including histone deacetylase 6 (HDAC6). We showed that Dido3-dependent targeting of HDAC6 is a key determinant of cilium size in growth-arrested cells. Dido3 availability at the centrosome governs ciliary HDAC6 levels, and redistribution of the two proteins controls tubulin acetylation. Increased Dido3ΔNT expression and interaction with HDCA6 results in ciliopathies. We used this mutation to interpret classical features of CIN, including frequency of chromosome breakage during cell growth. We consider that, by generating substrates for the non-homologous end-joining system, CIN-associated centromere fission helps to initiate breakage-fusion-bridge cycles and acquisition of additional copies of chromothriptic chromosomes.
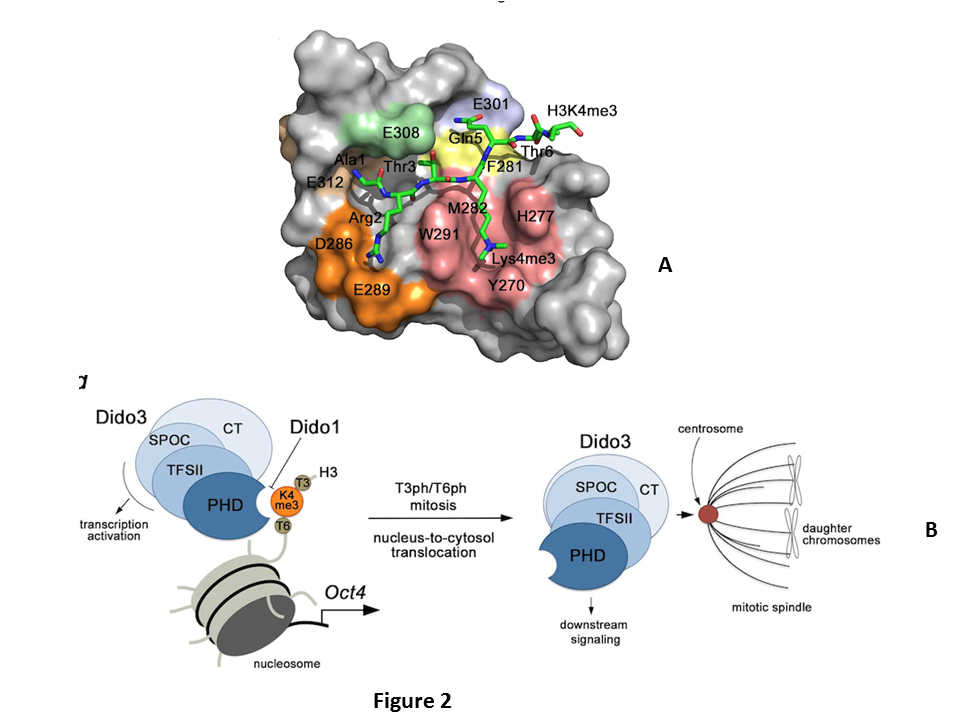
C-terminal truncation of the Dido3ΔCT gene causes early embryonic lethality. ESC derived from blastocysts of Dido3ΔCT mutants do not differentiate in vitro, in contrast to wild-type cells. Differentiation is restored by ectopic expression of wild-type Dido3 and constructs bearing different Dido domains, including the PHD domain. We showed that Dido3 PHD binding to histone H3K4me3 is disrupted by mitotic phosphorylation of an adjacent histone threonine, which triggers Dido3 translocation from chromatin to spindle microtubules. Our crystal structure of Dido3 PHD in complex with H3K4me3 revealed an aromatic-cage-like binding site. Biochemical, structural, and mutational analyses of the binding mechanism identified specificity and affinity determinants, and explained the affinity of other PHD domains for H3K4me3. Our findings link transcriptional control in embryonic development and regulation of cell division.