Francisco García del Portillo
Group Leader
Research summary
We hypothesize that the peptidoglycan that Salmonella remodels and synthetizes “inside the host cell” has a unique structure that plays a central role in the bacteria-host cell crosstalk.
Publications
SB Hernández, S Castanheira, MG Pucciarelli, JJ Cestero, G Rico-Pérez, A Paradela, JA Ayala, S Velázquez, A San-Felix, F Cava, F García-del Portillo. Peptidoglycan editing in non-proliferating intracellular Salmonella as source of interference with immune signaling. Plos Pathogens 2022; 18(1):e110241; DOI:
Huesa J, Giner-Lamia J, Pucciarelli MG, Paredes-Martínez F, García-del Portillo F, Marina A, Casino P. Structure-based analyses of Salmonella RcsB variants unravel new features of the Rcs regulon. Nucleic Acids Research 2021; 49(4):2357-2374. doi: 10.1093/nar/gkx1164.
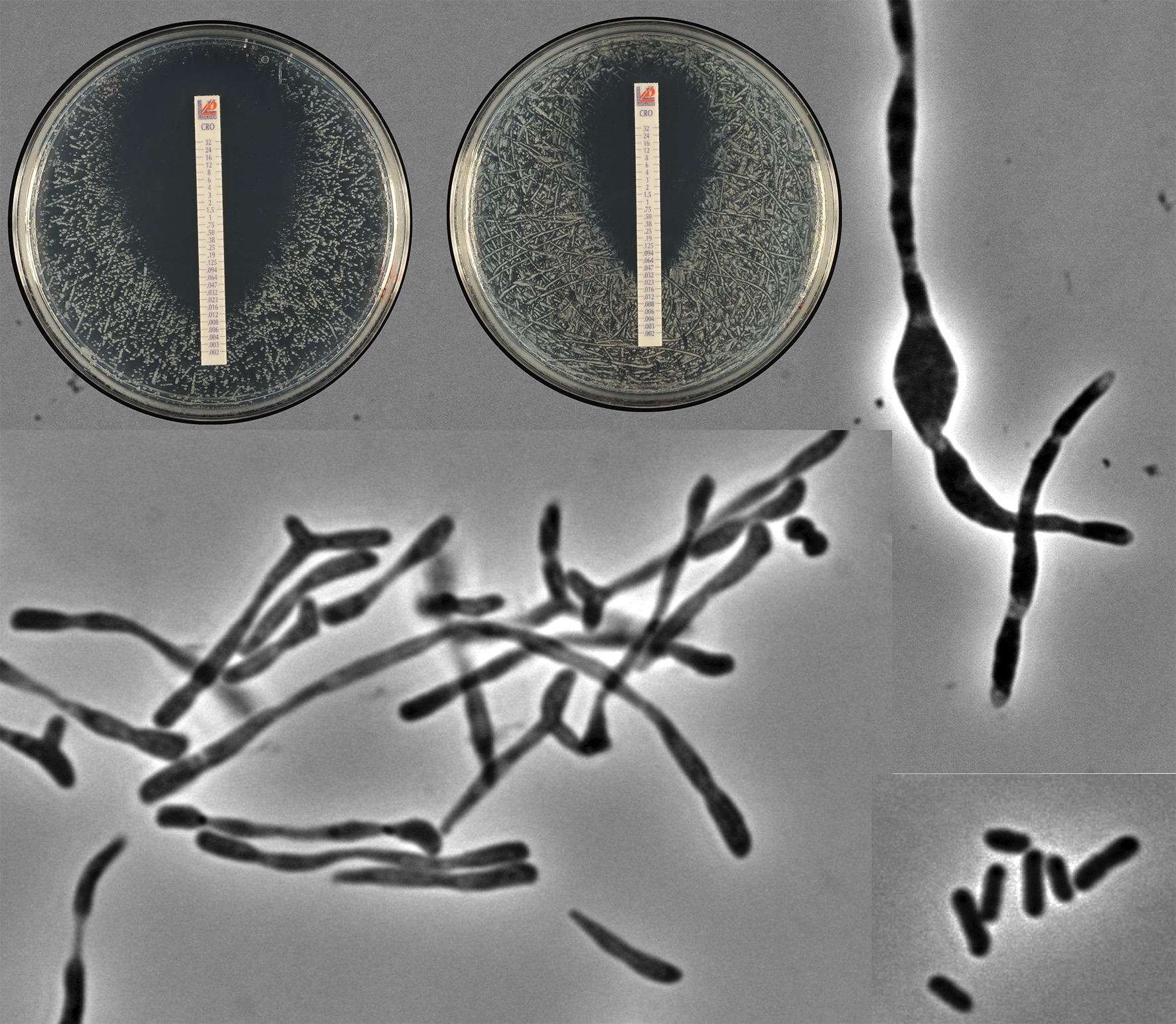
Castanheira S, López-Escarpa D, Pucciarelli MG, Cestero JJ, Baquero F, García-Del Portillo F. An alternative penicillin-binding protein involved in Salmonella relapses following ceftriaxone therapy. EBioMedicine 2020; 55:102771. adoi: 10.1016/j.ebiom.2020.102771
García-del Portillo. Building peptidoglycan inside eukaryotic cells: a view from symbiotic and pathogenic bacteria. Molecular Microbiology 2020; 113(3):613-626. doi: 10.1111/mmi.14452
Castanheira S, Cestero JJ, Rico-Pérez G, García P, Cava F, Ayala JA, Pucciarelli MG, García-Del Portillo F. A Specialized Peptidoglycan Synthase Promotes Salmonella Cell Division inside Host Cells. MBio. 2017 Dec 19;8(6)
Funding
|
|
Programa Nacional de cooperación Público-Privada (Subprograma INNPACTO) IPT-2012-0213-060000: Tecnologías innovadoras para el desarrollo de nuevos productos saludables a base de polifenoles (INNOFENOL) |
 |
The peptidoglycan (PG) is the main component of the cell wall and it was selected during the evolution of the domain Bacteria as macromolecule to withstand the turgor pressure and as platform to anchor surface appendages. Due to its unique physical and chemical properties as a single giant covalently-bound macromolecule, the PG is also the determinant that confers shape. Apart from these essential roles in the physiology of most bacteria, the PG modulates many host-pathogen interactions. It has molecular patterns that alarm our immune system and, in fact, a series of innate immune receptors senses PG fragments to initiate inflammation and combat the infection.
Our lab aims to decipher mechanistically how the PG impacts the course of an intracellular infection. The data obtained in our lab points to an opposite effect on immunity: changes in its structure triggered by cues perceived by intracellular bacteria inside the infected cell may promote immune evasion. This scheme is supported by single-cell studies that demonstrated the ability of Salmonella enterica to attenuate the NF-kB response to establish a persistent infection inside fibroblasts. We therefore hypothesize that the PG that is synthesized or remodeled de novo “inside the host cell” has a unique structure and plays a central role in the bacteria-host cell crosstalk.
Bottom-up approaches involving phenotypic analysis of mutants lacking defined enzymes involved in PG metabolism, structural analyses of the PG synthetized by intracellular bacteria and assays directed to measure activation of immune defenses, are currently used to determine the extent at which these structural changes in the PG responding to intracellular cues contribute to restrain growth and facilitate the persistent infection. Other areas of interest include the evolution of enzymes linked to PG metabolism and enzymes encoded exclusively by genomes of some bacterial pathogens that are up-regulated during the intracellular infection. These pathogen-specific enzymes are currently characterized to develop novel therapies targeting cell wall metabolism selectively inside the eukaryotic cell.









