Marta Nieto
Group Leader
Research summary
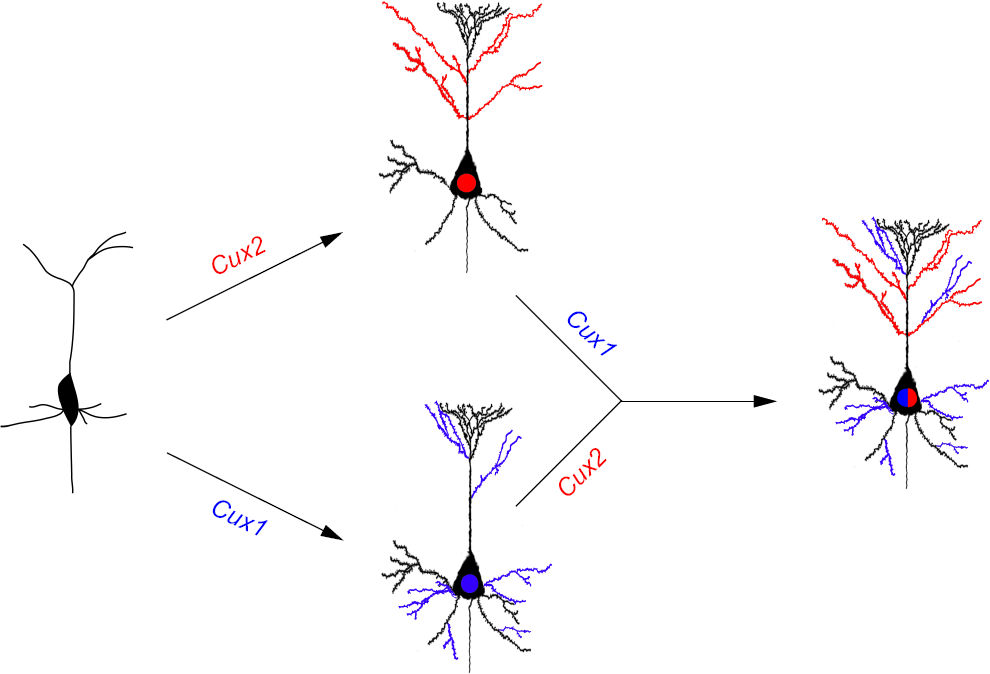
Our most recent work, using knockout and knockdown studies combined with morphological, molecular and electrophysiological analysis, demonstrates that Cux1 and Cux2 are intrinsic and complementary regulators of dendrite branching, spine development, and the functional synapse of neurons of layer II-III of the cerebral cortex.
Publications
De León Reyes NS, Bragg L, Nieto M. The development of the corpus callosum. Development 2020; 147: dev189738
De León Reyes NS, Mederos S, Varela I, Weiss LA, Perea G, Galazo MJ, Nieto M Transient callosal projections of L4 neurons are eliminated for the acquisition of local connectivity. Nat Commun 2019 Oct 7;10(1):4549.doi:10.1038/s41467-019-12495-w.
Weiss LA and Nieto M. The crux of Cux genes in neuronal function and plasticity. Brain Res 2018 Mar 6. pii: S0006-8993(18)30114-8.
Briz CG, Navarrete M, Esteban JA and Nieto M. In utero electroporation approaches to study excitability of neuronal subpopulations and single cell connectivity. J. Vis. Exp. 2017, (120), e55139.
Wang CF, Hsing HW, Zhuang ZH, Wen MH, Chang WJ, Briz CG, Nieto M, Shyu BC, and Chou SJ. Lhx2 expression in postmitotic neurons initiates the assembly of the thalamocortical somatosensory circuit. Cell Rep. 2017 Jan 24; 18(4):849-856.
Doan RN, Bae B, Johnson MB, Cubelos B, Chang C, Hossain AA, Al-Saad S, Mukaddes NM, Oner O, Al-Saffar M. The Homozygosity Mapping Consortium for Autism, Nieto M, Walsh CA. Mutations in Human Accelerated Regions Disrupt Cognition and Social Behavior. Cell 2016 Oct 6;167(2):341-354.e12.
 In our studies, we aim to define the cellular and molecular mechanisms that govern the generation of neurons and circuits of the mammalian cerebral cortex. The mammalian cerebral cortex, responsible for most aspects of cognition and behaviour, is the most recently evolved structure in the human brain. A large number of functionally and morphologically distinct neuronal types specify brain cortical areas and control cerebral functions. We help to understand the programs that specify the identity of the neurons in the upper layers of the cerebral cortex. This subpopulation of pyramidal neurons characterises higher mammals and is expanded in humans, probably contributing to the increased cognitive capacity of the mammalian brain. It is the last to appear during development and in evolution. Our research showed that the transcription factors Cux1 and Cux2 are responsible for the extremely high degree of connectivity of these neurons and their participation in intra-cortical circuits that control higher brain functions. In our ongoing work, we dissect the neuronal characteristics modified by these genes to generate specialised neurons. We analyse how these are coordinated with experience and plasticity to generate the stereotyped networks of the human brain.
In our studies, we aim to define the cellular and molecular mechanisms that govern the generation of neurons and circuits of the mammalian cerebral cortex. The mammalian cerebral cortex, responsible for most aspects of cognition and behaviour, is the most recently evolved structure in the human brain. A large number of functionally and morphologically distinct neuronal types specify brain cortical areas and control cerebral functions. We help to understand the programs that specify the identity of the neurons in the upper layers of the cerebral cortex. This subpopulation of pyramidal neurons characterises higher mammals and is expanded in humans, probably contributing to the increased cognitive capacity of the mammalian brain. It is the last to appear during development and in evolution. Our research showed that the transcription factors Cux1 and Cux2 are responsible for the extremely high degree of connectivity of these neurons and their participation in intra-cortical circuits that control higher brain functions. In our ongoing work, we dissect the neuronal characteristics modified by these genes to generate specialised neurons. We analyse how these are coordinated with experience and plasticity to generate the stereotyped networks of the human brain.
We also identified molecular mechanisms of axonal modelling and plasticity linked to these neurons, which participate in formation and physiology of brain circuits. In collaboration with other CNB groups, we study models of cell migration. Our research provides basic knowledge of the mechanisms of neural specification and circuit formation, the potential programs of reprogramming neurons, and the specific advantages and plasticity of the human brain. These data have broad, direct implications for understanding the specific functions of the cortex in intellectual processing. They might also explain underlying mechanisms of brain diseases that originate in childhood, and those of neurodegeneration, which is increasingly reported as plasticity-related.