Dimitrios Balomenos
Group Leader
Research summary
Apoptosis is considered a basic mechanism for limiting T cell memory expansion known as homeostasis. Nevertheless, we find that the control of activation of memory T cells and of proliferation are also fundamental for memory T cell homeostasis. We suggest that memory responses and homeostasis require a combination of control in activation, apoptosis and proliferation.The cell cycle inhibitor p21 suppreses autoimmunity and controls T cell memory responses.
Publications
Balomenos D, Shokri R, Daszkiewicz L, Vazquez-Mateo C, Martinez-A C. On how Fas apoptosis-independent pathways drive T cell hyperproliferation and lymphadenopathy in lpr mice. Front Immunol 2017; 8: 237.
Rodríguez-Frade JM, Guedán A, Lucas P, Martínez-Muñoz L, Villares R, Criado G, Balomenos D, Reyburn HT, Mellado M. Use of lentiviral particles as a cell membrane-based mFasL delivery system for in vivo treatment of inflammatory arthritis. Front Immunol 2017; 8: 460.
Rackov G, Shokri R, Alvarez De Mon M, Martinez-A. C, Balomenos D. The role of IFN-beta during the course of sepsis progression and its therapeutic potential. Front Immunol 2017; 8: 493.
Rackov G, Hernández-Jiménez E, Shokri R, Carmona-Rodríguez L, Mañes S, Álvarez-Mon M, López-Collazo E, Martínez-A C, Balomenos D. p21 mediates macrophage reprogramming through regulation of p50-p50 NF-κB and IFN-β. J Clin Invest 2016; 126:3089-103
Daszkiewicz L, Vázquez-Mateo C, Rackov G, Ballesteros-Tato A, Weber K, Madrigal-Avilés A, Di Pilato M, Fotedar A, Fotedar R, Flores JM, Esteban M, Martínez-A C, Balomenos D. Distinct p21 requirements for regulating normal and self-reactive T cells through IFN-gamma production. Sci Rep 2015; 5:7691
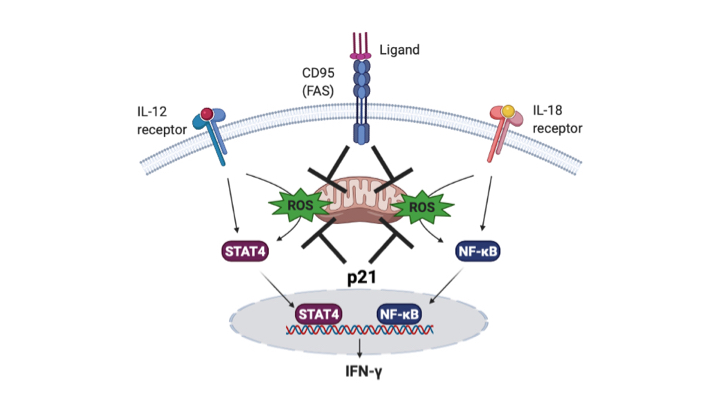
Increased immune responses and hyperinflammation govern the development and progression of diseases that extend from Autoimmunity to COVID-19. In order to neutralise inflammatory responses, the immune response needs to be supressed. Alternatively, in cancer, immunosuppressed immunity requires reactivation. Therefore is it essential to understand systems that regulate these responses. Notably, our work points to p21 as a regulator of the balance between hyperactivation and immunosuppression by controlling mitochondrial Reactive Oxygen Species (mROS). Our recent work shows that mROS is essential for IFN-gamma production by memory T cells after IL-12 plus IL-18 challenge (Rackov et al 2020). IFN-gamma orchestrates inflammatory responses in inflammation-induced diseases. Remakably, Fas controls mROS and IFN-gamma induction independently of its apoptosis inducing potential (Figure 1). Our current work (in preparation) indicates that p21 modulates mROS and IFN-gamma production by memory T cells, corroborating our published data, showing that p21 overexpression tempers autoreactive T cells and IFN-gamma production (Daszkiewicz et al, 2015). Therefore, high expression of p21 lowers T cell overactivity, while lack of p21 enhances responses by regulating mROS production (Figure 1).
Similarly to memory T cells, p21 regulates the inflammatory potential in macrophages. We have shown a dual regulatory role for p21; first, in macrophage activation to M1 state (Trakala et al, 2009 ) and, second, in macrophage reprogramming from M1 to the M2 unresponsive state. Lack of p21 prevents macrophage reprogramming to M2 status (Rackov G et al, J Clin Invest 2016). Our present results firmly show that mROS, which is regulated by p21, is an early regulator of the inflammatory response of M1 macrophages as it enhances M1 responses as early as five minutes post-activation, and leads to NF-kB activation and ultimately to inflammatory cytokine production. The direct interaction of p21 and mitochondria in M1 macrophages is shown in Figure 2.
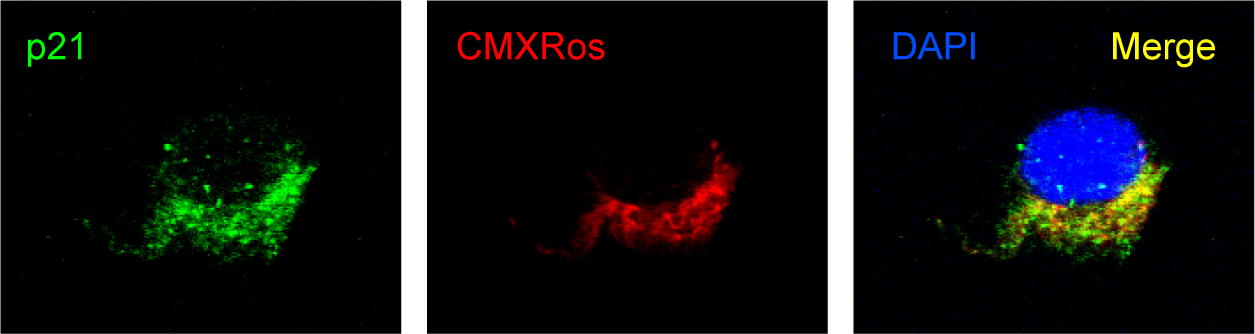
Figure 2. Confocal microscopy of stimulated macrophages shows direct interaction of p21 with mitochondria specifically stained by CMXRos. Polarisation of mitochondria is evident due to their activated status of macrophages.








