Adrian Valli
Ramón y Cajal researcher
Research summary
It is worrisome the negative impact of pathogens over crop yield, with losses of roughly 30% of global productivity. In the laboratory, we study viral plant pathogens with the aim of understanding how they cause disease, and also discovering networks of plant proteins that act as defensive barriers. These studies not only help us to expand our knowledge about plant/virus co-evolution, but also allow us to design more and better strategies to provide plants with an enhanced antiviral resistance.
Publications
Valli A, García Lopez R, Ribaya M, Martínez FJ, García Gómez D, García B, Gonzalo I, Gonzalez de Prádena A, Pasin F, Montanuy I, Rodríguez-Gonzalo E, García JA. Maf/ham1-like pyrophosphatases of non-canonical nucleotides are host-specific partners of viral RNA-dependent RNA polymerases. PLOS Pathogens 2022; 18(2): e1010332.
González de Prádena A, Sánchez Jiménez A, San León D, Simmonds P, García JA, Valli AA. Plant virus genome is shaped by specific dinucleotide restrictions that influence viral infection. MBio 2020; 11: e02818-02819
Qin L, Shen W, Tang Z, Hu W, Shangguan L, Wang Y, Tuo T, L Z., Miao W, Valli A, Wang A, Cui H. A Newly Identified Virus in the Family Potyviridae Encodes Two Leader Cysteine Proteases in Tandem That Evolved Contrasting RNA Silencing Suppression Functions. Journal of Virology 2020; 95(1). e01414-20
Ochoa J, Valli A, Martín-Trillo M, Simón-Mateo C, García JA, Rodamilans B. Sterol isomerase HYDRA1 interacts with RNA silencing suppressor P1b and restricts potyviral infection. Plant Cell Environ 2019; 42: 3015-3026
Canto-Pastor A, Santos B, Valli A, Summers W, Schornack S, Baulcombe DC. Enhanced resistance to bacterial and oomycete pathogens by short tandem target mimic RNAs in tomato. Proc Natl Acad Sci U S A 2019; 116, 2755-2760.
Funding
Uncovering the mechanism and potential uses of viral RNA polymerase slippage. Spanish MICINN (BIO2015-73900-JIN), from 2017 to 2019 (3 years).
Ramón y Cajal Program (RYC2018-025523-I). Spanish MICINN, from 2020 to 2024 (5 years)..
The paradigm of Euphorbiaceous-Virus interaction: a multidisciplinary approach to develop novel antiviral strategies in plants. Spanish MICINN (PID2019-110979RB-I00), from 2020 to 2022 (3 years).
The Euphorbiaceous-Virus Coevolution. Spanish Research Council (CSIC), from 2020 to 2022 (3 years).
Food security is under threat due to the increasing world population and the hampering effects of global warming over crop production. In this context, it is particularly worrisome the additional negative impact of pathogens over crop yield, with losses of roughly 30% of global productivity. Therefore, gaining insight about plant pathogens is critical to reveal how they cause disease and to discover plant protein networks acting as defensive barriers. These basic studies not only help to expand our knowledge about plant/pathogen co-evolution, but also allow the design of more and better strategies to provide plants with an enhanced resistance.
Our laboratory studies plant/virus co-evolution by paying special attention to RNA viruses of the Potyviridae family –the biggest and most relevant group of plant RNA viruses. For that, we follow a multidisciplinary approach, which include (i) synthetic biology to build and manipulate viral infectious clones; (ii) genomics/transcriptomics studies aiming to decipher the effects of plants over viruses, and vice versa; (iii) structural studies by cryo-electron microscopy to define 3D structures of relevant viral proteins, (iv) metabolomics studies by HPLC-MS/MS to untangle plant physiological changes due to viral diseases, and (v) viral ecology to understand the interaction occurring between plant and viruses in the field.
Our recent findings include (i) the restriction of UpA dinucleotides in potyviral genomes and the associated cost when viruses break such a restriction; (ii) the host-specific role of viral-derived ITPases during viral infection; (iii) the potential antiviral activity of plant non-canonical nucleotides in the cytoplasm of infected cells; and (iv) the key role of viral-derived proteins with AlkB domains of releasing epigenetics marks associated with RNA degradation from viral genomes.
Importantly, we have a fruitful network of collaborators in Spain (Madrid and Barcelona) and abroad (France, UK, China, and Argentina).
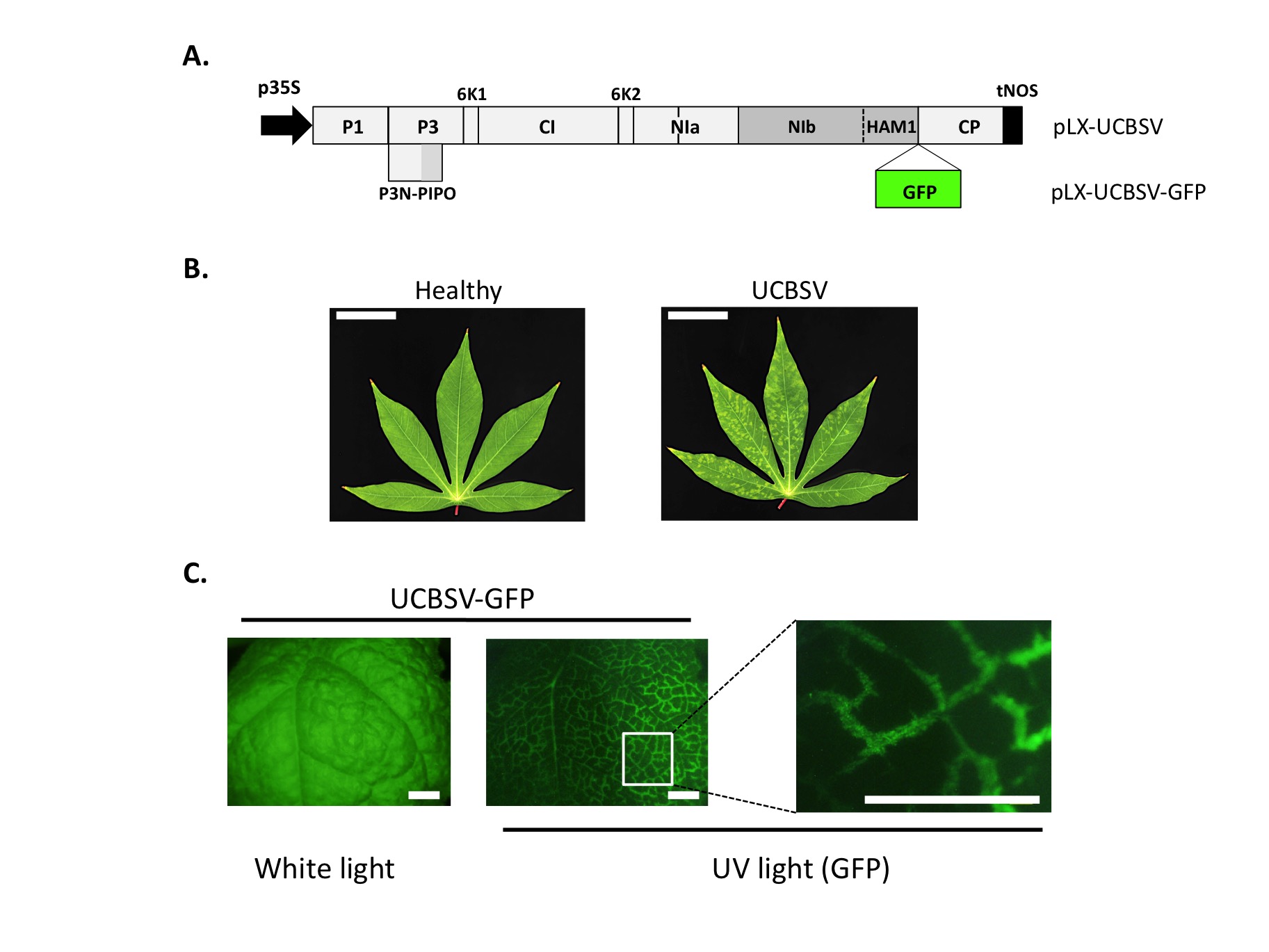
Figure Legend: Infection of plants with laboratory-designed cDNA clones of UCBSV. (A) The RNA genome of UCBSV was retrotranscribed to cDNA and this sequence was then inserted in between a strong promoter (p35S from Cauliflower mosaic virus) and a terminator (tNOS from Agrobacterium nopaline synthase) to generate the infectious cDNA plasmid termed pLX-UCBSV (Pasin et al., 2017, ACS Synthetic Biology). This plasmid was further modified to tag the virus with GFP, thus generating pLX-UCBSV-GFP. (B) Leaves of cassava plants (Manihot esculenta) at 2 months post-inoculation observed under white light (bar = 4 cm). Symptoms of infection are clearly detected in upper leaves of plants inoculated with pLX-UCBSV. (C) One leaf of a Nicotiana benthamiana plant, infected with UCBSV-GFP, observed under white light and UV light (to detect GFP-derived fluorescence) with a stereomicroscope. GFP is distributed across primary and secondary veins as the virus spread through the leaf (bar = 0.5 cm).


 Laboratory Members
Laboratory Members



