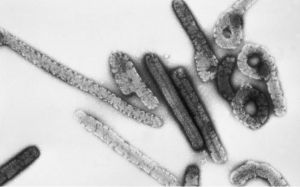
So far only supportive treatment
To date, there are no effective antiviral drugs or vaccines, and Marburg virus disease can only be treated supportively. Therefore, any epidemic causes unacceptable suffering in the affected countries and has the potential to further spread the virus. Thus, development of treatments or vaccines are needed.
Innovative vaccine development and preclinical test pipeline
Two vaccines against Marburg viruses are currently in Phase I clinical trials. However, they only target a single antigen of the virus. This could impair their protective effect. Thus, exploring new strategies, the MARVAX consortium will develop novel vaccine candidates based on two different viral vectors that will express multiple Marburg virus antigens. Both combined approaches are expected to maximise the immunogenicity and protective effect of the vaccine candidates.
Above all, the MARVAX consortium is joining forces with experts in vaccine development and production, virology, and immunology to go significantly beyond previous research approaches. The novel vaccine candidates against Marburg viruses will be generated by the CSIC National Centre for Biotechnology (CNB-CSIC) and the Institut Pasteur, and their immunogenicity and efficacy will be evaluated in animal models by BNITM, CNB-CSIC and Institut Pasteur in close collaborationCZ vaccines will develop the manufacturing process and generate clinical batches of the best-in-class vaccines that will be ready for future phase I clinical trials.
"I am very pleased that we were able to convince the European Commission with our study concept," says Prof. César Muñoz-Fontela, head of the Virus Immunology research group at the BNITM and MARVAX project coordinator. "Marburg virus outbreaks are becoming more frequent and more and more countries are affected. We must finally develop effective vaccines in order to contain outbreaks at an early stage with the help of ring vaccinations, as we did with Ebola, and put an end to the deaths!"
Juan García-Arriaza, CNB-CSIC researcher, highlights the value of their participation in this international collaboration. "Our vaccine development platform will allow us to generate new and promising vaccine candidates against this virus. We hope that they will be able to robustly activate the immune system and protect against infection, which will allow us to select the best candidates for future evaluation in clinical trials."
Eugenia Puentes, Head of R&D at CZ Vaccines, said: "We are committed to ensuring that clinical batches are generated under GMP conditions, as this is a critical step in advancing clinical development and future vaccine production.
More About MARVAX platfform
The MARVAX study platform will receive a Horizon Europe grant of 7.4 million euros from the European Framework Programme for Research and Innovation over the next four years. The MARVAX consortium includes the Institut Pasteur (France), the Spanish National Research Council (Spain), the BNITM (Germany) and CZ Vaccines (Spain). The platform studies included in the MARVAX consortium will be carried out in close co-operation with the WHO. This should also ensure that a rapid response is possible in the event of renewed Marburg virus outbreaks.
Joint Press Release issued by the Communications Offices from the MARVAX consortium members
BNTIN, Pasteur Institute, CZ vaccines-Zendal Group, CNB & CSIC