Ana C. Carrera
Group Leader
Research summary
The focus of the team´s work is to define cancer progression drivers with special emphasis in PI3-kinase-and NRF2-controled pathways and focusing in lung squamous cell carcinoma, currently without an effective targeted therapy
Publications
Pekarek L, De la Torre-Escuredo B, F.-Martinez O, G.-Montero C, Saez MA, C-Prieto D, Guijarro LG, Saz JV, De Castro-Martinez P, T.-Carranza D, Pekarek T, Carrera AC, Alvarez-Mon M, Ortega MA. Towards the Search for Potential Biomarkers in Osteosarcoma: State-of-the-Art and Translational Expectations. Int J Mol Sci. 2022, 23(23):14939.
González-García A, Garrido A, Carrera AC. Targeting PTEN Regulation by Post Translational Modifications. Cancers (Basel) 2022, 14(22):5613.
Yadav S, Garrido A, Hernández MC, Oliveros JC, Pérez-García V, Fraga MF, Carrera AC. PI3Kβ-regulated β-catenin mediates EZH2 removal from promoters controlling primed human ESC stemness and primitive streak gene expression. Stem Cell Reports 2022, 17(10):2239-2255.
Olazábal-Morán M, Sánchez-Ortega M, Martínez-Muñoz L, Hernández MC, S. Rodríguez M, Mellado M and Carrera AC. Fluctuations in AKT and PTEN Activity Are Linked by the E3 Ubiquitin Ligase cCBL Cells 2021, 10(11):2803.
Sánchez-Ortega M, Carrera AC and Garrido A. Role of NRF2 in Lung Cancer. Cells 2021, 10, 1879.
Olazabal-Morán M, González-García A, Carrera AC. Functions of Nuclear Polyphosphoinositides. Handb Exp Pharmacol 2020; 259: 163-181
Vallejo-Díaz J, Chagoyen M, Olazabal-Morán M, González-García A, Carrera AC The Opposing Roles of PIK3R1/p85α and PIK3R2/p85β in Cancer. Trends Cancer 2019; 5(4):233-244.
Carrera AC & Anderson R. The cell biology behind the oncogenic PIP3 lipids.J Cell Sci 132 2019; jcs228395.
Funding



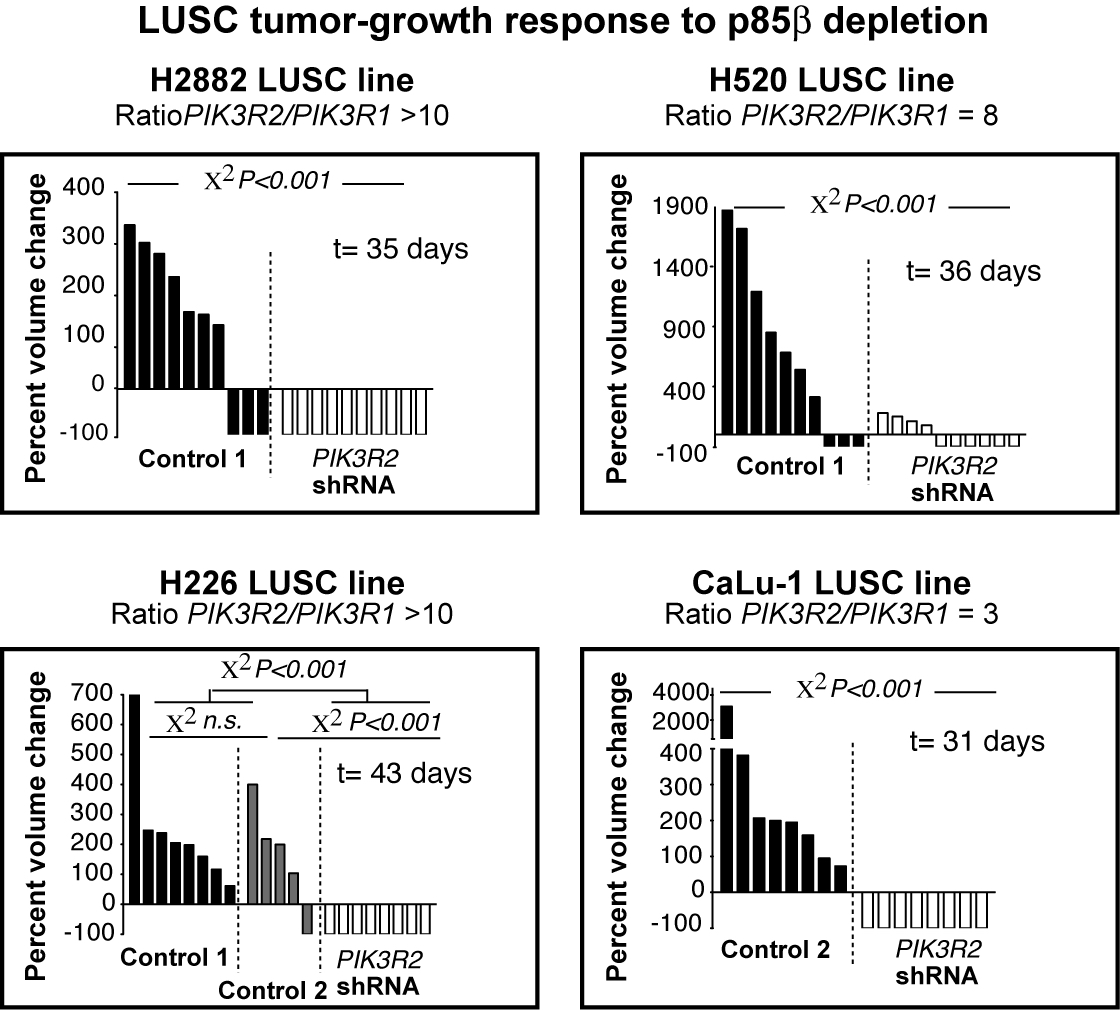
1) Inhibitory compounds for LUSC treatment based on interference with PIK3R2.
Lung cancer is the most common cause of cancer death worldwide. Approximately 85% of the lung cancer patients exhibit a histological subtype known as non-small cell lung carcinomas (NSCLC). This cancer type comprises several subtypes including adenocarcinoma (LUAD) and lung squamous cell carcinoma (LUSC) etc. Advances in basic and clinical research have increased our understanding of the mechanisms inducing cancer progression, leading to development of pharmacological inhibitors that block specific signaling pathways and can be used for cancer treatment (targeted therapies). In NSCLC this approach has been successfully used in LUAD tumors, with LUSC tumors still waiting for an efficient targeted therapy.
Different experimental approaches developed over last year’s support that p85b (encoded by PIK3R2) acts as a tumor driver in LUSC cancer (PMID: 27835880). Indeed, LUSC samples exhibit a frequent increase of p85b levels (PMID: 30961830), and at least in melanoma p85b increase correlate with tumor grade (PMID: 25217619). The idea of this project is to capitalize this knowledge for the screening and development of inhibitory compounds for LUSC based on interference with PIK3R2 action.
2) PIP3 actions in TUMOR microenvironment: hypoxia
In the course of our studies on the growth of human tumours as xenografts in mice, we noticed that interference with the action of PI3-kinases alters the response of cells to oxygen deprivation. Since solid tumours often grow in low oxygen conditions (hypoxia), we are investigating the crossroads between PIP3 regulation and the expression of the HIF transcription factors that mediate cancer cell adaptation to low oxygen levels.
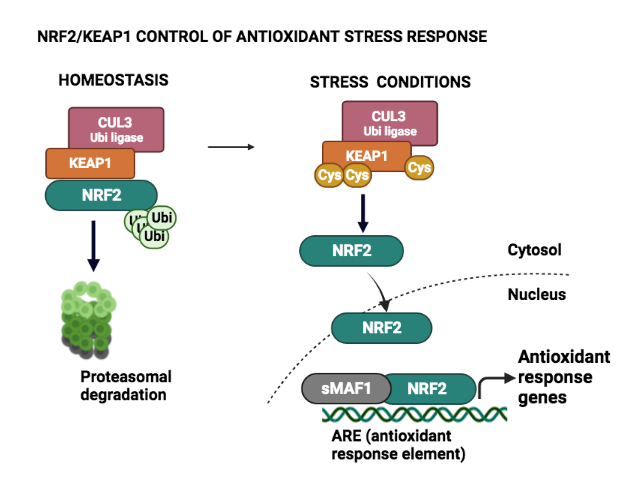
3) NRF2 regulated pathways as a source for novel LUSC therapeutic targets
NFE2L2 (NRF2) is a transcription factor regulated by KEAP1 (KEAP1), NRF2 directs the response of the cells to reactive oxygen species (ROS). In the last decade, the introduction of next-generation sequencing (NGS) has allowed the description of the mutational profile of different tumors. One of the relatively high frequency gene mutations of lung squamous cell carcinoma (LUSC), are those affecting NFE2L2/KEAP1. The first aim of this project is to define whether this pathway acts as a tumor driver for squamous LUSC cancer. Second, to define NFE2L2/ KEAP1 downstream targets (using CRISPR/Cas9) and finally to test if these hits may regulate LUSC growth and can be considered novel therapeutic targets for LUSC.