An international team, in which researchers from the Centro Nacional de Biotecnología (CNB) of the CSIC and the Universidad Autónoma of Madrid (UAM) participated, has discovered the existence of a considerable number of miniproteins encoded by the genome of the bacterium Listeria monocytogenes. The study, published in the journal Nature Microbiology, shows that one of these proteins, termed Prli42, plays a key role in receiving environmental signals.
The finding, by a group led by scientist Pascale Cossart of the Pasteur Institute in Paris, offers an opportunity to study these miniproteins and their possible biotechnological applications. In addition, it helps to understand the mechanism by which the stressosome macrocomplex is anchored to the bacterial membrane, a process necessary for receiving signals from the environment and alerting the bacteria in stress situations.
"To adapt to changing environments, throughout their evolution bacteria have developed many functions that activate stress response genes. In some Gram-positive bacteria, the stressosome macrocomplex in the cytoplasm transmits signals and activates functions that alert the bacterium and allow it to survive," says Francisco García-del Portillo, a CSIC scientist at the at the Centro Nacional de Biotecnología.
"The stressosome structure is well-studied in some Gram-positive bacteria such as Bacillus subtilis, but it is not yet known how they capture signs of stress," says María Graciela Pucciarelli, a researcher of the Universidad Autónoma of Madrid at the Center for Molecular Biology Severo Ochoa.
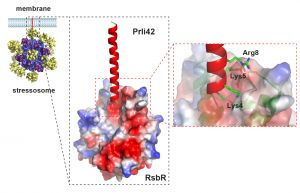
The scientists used a new proteomic approach in the bacterium Listeria monocytogenes, a human pathogen, which led to the discovery of 19 internal translation start sites (in protein-coding regions) and six miniproteins encoded in genes previously associated exclusively with small RNA molecules. Among these is the miniprotein Prli42, which is conserved in Firmicutes (a group of Gram-positive bacteria). The miniprotein is anchored to the bacterial membrane and facilitates stressosome binding.
The study offers an important technical and conceptual advance regarding the existence of miniproteins, whether encoded in regions previously cataloged as intergenic or due to the presence of internal translation initiation sites within coding regions. “The application of this type of proteomic approach in other organisms will undoubtedly bring new surprises, in anticipation of an important and previously unsuspected biological role for miniproteins," adds García-del Portillo.
The study also analysed the way the stressosome macrocomplex associates with the bacterial envelope, and thus enters into contact with still-unknown environmental signals.
Source: CSIC
- Francis Impens, Nathalie Rolhion, Lilliana Radoshevich, Christophe Bécavin, Mélodie Duval, Jeffrey Mellin, Francisco García del Portillo, M. Graciela Pucciarelli, Allison H. Williams, Pascale Cossart. N-terminomics identifies Prli42, a membrane miniprotein critical for stressosome activation in Listeria monocytogenes. Nature Microbiology. DOI: 10.1038/nmicrobiol.2017.5