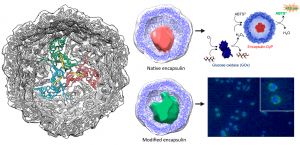
Some bacterial species have small internal protein capsules in which certain reactions take place. These compartments, termed encapsulins, act as bacterial semiorganelles.
Scientists at the Centro Nacional de Biotecnología of the CSIC (CNB-CSIC), in collaboration the University of Twente (The Netherlands), have now described the structural characteristics of these nanometric cages. The results suggest that encapsulins could be used as therapeutic agents, biosensors, bioreactors, or have other nanotechnological applications. The study was published in the journal ACS Nano.
The authors used electron cryomicroscopy techniques to decipher the three-dimensional structure of encapsulins. In addition, they showed that encapsulins are robust, stable, and functional both in vitro and in vivo. "We found that encapsulins immobilized on a surface are able to act as bioreactors. We also observed that they are internalized by cells in culture, which makes them candidate vehicles for therapeutic agents in controlled drug release," explains José R. Castón, CNB-CSIC researcher and author of the study.
One of the most important characteristics of encapsulins is their porosity. Tiny holes on their surface (a few nanometers in diameter) allows smaller molecules to enter and exit freely. The largest proteins are confined in the interior, where they carry out specific reactions.
Utility as biosensors and as drug vehicles
To study their function as nanoreactors, the scientists immobilized the encapsulins on a glass surface. After addtion of appropriate substrates to the medium, the protein trapped within retained its ability to carry out the appropriate reaction.
For the second experiment, encapsulins bearing an easily detectable fluorescent protein were used. When cultured together with macrophages, the encapsulins were internalized by the cells.
Although virus capsids and other protein nanostructures are already widely studied as vehicles or bioreactors, this study is one of the first to focus on encapsulins. "We know very little about them. It is thought that they might have originally been viruses that were internalized and incorporated by the bacteria. Or vice versa– the encapsulins could have been the origin of the viruses,” explains Castón. “This study is the first probe of their structure and possible applications".
.
- Putri RM, Allende-Ballestero C, Luque D, Klem R, Rousou KA, Liu A, Traulsen CH, Rurup WF, Koay MST, Castón JR, Cornelissen JJLM. Structural Characterization of Native and Modified Encapsulins as Nanoplatforms for in Vitro Catalysis and Cellular Uptake. ACS Nano. 2017 Dec 1. doi: 10.1021/acsnano.7b07669.