“Bacteriophages are viruses that infect bacteria. In addition to their interest as a biological model because of their genetic simplicity but great structural complexity, they are now a new focus of attention for researchers given their potential as an alternative to the use of antibiotics, ”explains José L. Carrascosa, a researcher at the National Center for Biotechnology and co-director of work.
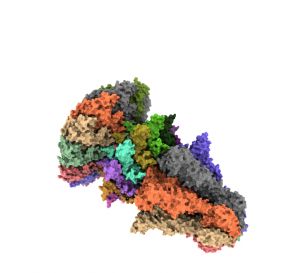
During the infection cycle of T7, a viral capsid is formed where the virus needs to include its genetic material, introducing it through a small entrance channel formed by the portal protein that is subsequently closed with the virus's tail protein. Until now it was unknown which was the mechanism that allowed to open and close this "door" in a controlled manner.
Ana Cuervo, first author of the work, together with Montserrat Fàbrega-Ferrer from IBMB and the IRB Barcelona, highlights how “cryoelectron microscopy and its complementation with crystallography techniques allow not only to solve the atomic structure of large complexes that pose a technical challenge, but also to define a model of opening and closing the portal during the process of translocation of the viral DNA ”. Using this approach, researchers have been able to identify the interactions between the different proteins that allow changes in the conformation of the complex, from an open structure to allow the passage of viral DNA to a closed one when forming mature viral particles.
This work has been funded by “Severo Ochoa” and “María de Maeztu” excellence programs, the “Ramón y Cajal” program of the Ministry of Science, Innovation and Universities, and the European iNEXT and Instruct-ERIC projects.
More information
Ana Cuervo, Montserrat Fàbrega-Ferrer, Cristina Machón, José Javier Conesa, Francisco José Javier Fernández, Rosa Pérez-Luque, Mar Pérez-Ruiz, Joan Pous, María Cristina Vega, José L. Carrascosa y Miquel Coll. Structures of T7 bacteriophage portal and tail suggest a viral DNA retention and ejection mechanism. Nature Communications. DOI: 10.1038/s41467-019-11705-9