Researchers at the Centro Nacional de Biotecnología of the CSIC (CNB-CSIC) have shown, in collaboration with scientists from the US and Australia, the intricacy of the interactions between the membrane proteins responsible for transmitting an activation message to cells of the immune system.
The study, published in the journal Proceedings of the National Academy of Sciences USA, analyses complexes formed by a membrane receptor protein able to detect external signals and an adapter molecule that translates the information and transmits it to the cell interior. This assembly is produced through the transmembrane domains of both proteins.
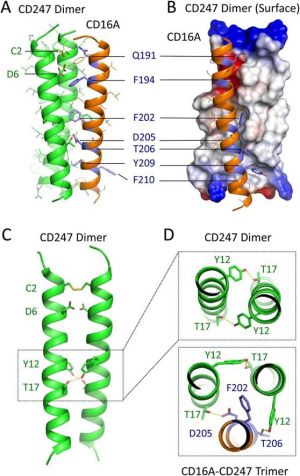
"The best-known mechanism to date is based on interaction between an acid and a basic amino acid, a system that involves a single amino acid from each subunit of the complex,” explains Alfonso Blázquez, CNB-CSIC scientist and lead author of the study. “Here we describe for the first time that there is a second mode of assembly based on a network of several aromatic and polar amino acids."
The scientists specifically studied the complex formed by the CD16A receptor with two adapter molecules (FceR1g and CD247). Since both subunits of the complex have acidic amino acids, there must be a different association mechanism. Among the results of this work, the authors also defined the amino acids involved in receptor degradation and renewal, as well as in its presence at the plasma membrane.
The study shows that throughout evolution, different systems have developed for cell receptor interaction with other transmembrane proteins to form complexes that can signal activation to the leucocyte interior. "It could be argued that, from an evolutionary viewpoint, this interaction system is more robust than the one we already know, since complex formation does not depend on a single, absolutely critical amino acid," suggests Hugh Reyburn, director of the research team.
.
- Alfonso Blázquez-Moreno, Soohyung Park, Wonpil Im, Melissa J. Call, Matthew E. Call and Hugh T. Reyburn Transmembrane features governing Fc receptor CD16A assembly with CD16A signaling adaptor molecules. PNAS 2017; doi:10.1073/pnas.1706483114